White House releases after-action reviews on Afghanistan withdrawal
The documented lessons learned from the Afghanistan withdrawal became publicly available for the first time on Thursday with the White House’s release of its after action reviews. The review places…
Highlighting Icons: Anne Hathaway Fronts This Italian Fashion Brand's Latest Collection
Anne Hathaway has been chosen as the face of this brand’s newest “Icons” campaign, featuring a “timeless elegance with an edge.” Anne Hathaway is an actress who needs no introduction,…
Texas Senate Unanimously Passes Measure to End Complicity in Beijing’s Forced Organ Harvesting
A bipartisan measure has unanimously passed the Texas Senate as lawmakers advance steps to prevent their state from aiding in the Chinese regime’s forced organ harvesting. Senate Bill 1040, with…
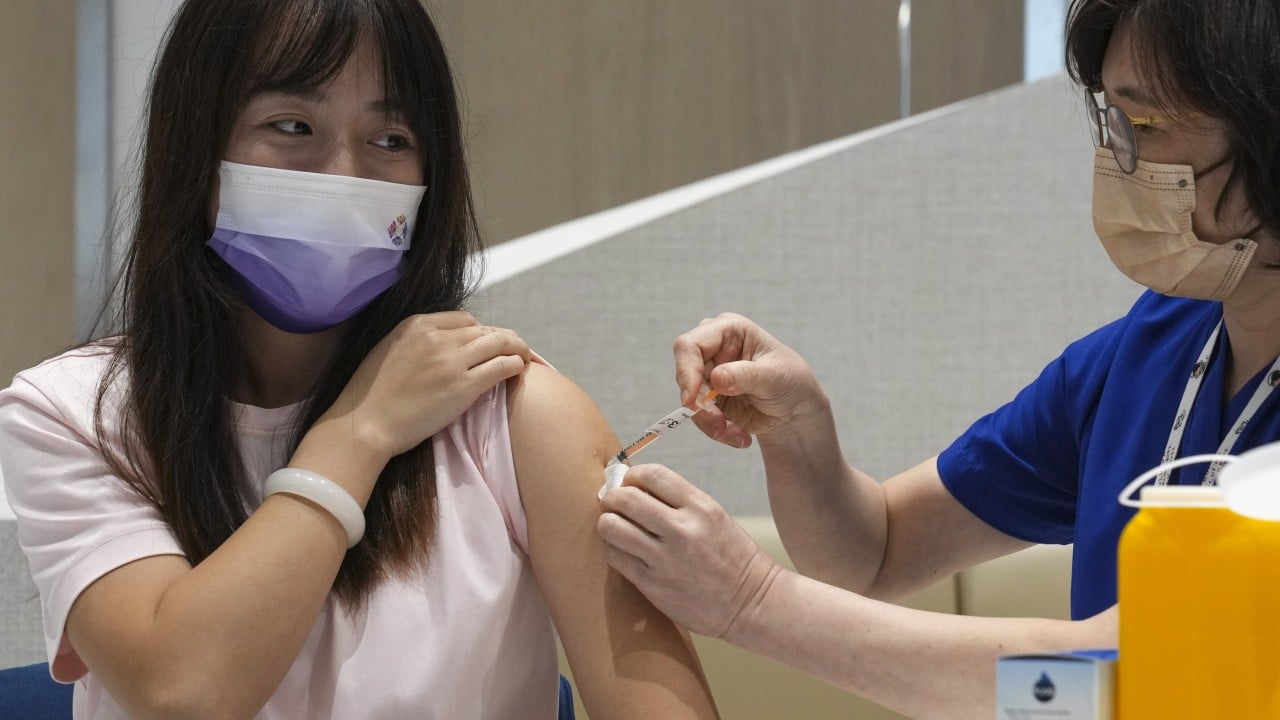
Covid vaccine rush in Hong Kong 2 weeks before jabs become chargeable
Hong Kong doctors observe Covid vaccine rush with 2 weeks to go before jabs become chargeable Health sector representatives estimate costs could be similar to what mainland visitors are charged…
The Macallan James Bond 60th Anniversary Release: Celebrating Excellence
The Macallan James Bond 60th Anniversary Release is a magnificent collection of whiskies to vicariously live a spy life. The Macallan James Bond 60th Anniversary Release at Harrods London. Image:…
Recession fears rise as growth projections are cut and exports continue to decline in 2023 – Thai Examiner
Thailand’s current account in the last three years tells the story of an economy that has seen a structural change since the closure of the kingdom in April 2020. This…
Report Shows Gen Z, Millennials Dominate US, Canadian Cannabis Sales
With cannabis on the rise, it’s fair to say that the cannabis consumer landscape has shifted immensely. While there may still be that prevailing idea of the typical cannabis user…
Making a military move? The road may be bumpy; these tips will help
As the moving season — from May to September — gear ups for service members and families, persistent problems with labor shortages make it difficult to predict what those moves…
A Wondrous Easter Feast at The Line Shangri-La Hotel Singapore
A Wondrous Easter Feast at The Line Easter is a time to celebrate new beginnings and share joy with your loved ones. If you are looking for a wondrous feast…
Migrant workers in fishing industry continue to face decent work deficit
Migrant workers in the fishing and seafood processing industry in Thailand continue to face major decent work deficits, despite the major reforms government has introduced, according to Benjamin Harkins, Technical…